
CMSI Phase III:
Precompetitive Collaboration on Safety Research & Methods Development
Collaboratively planning and executing cultured meat safety research.
To advance cellular agriculture, it is critical to create public information to guide evidence-based policymaking and regulatory decisions.
In Phase I, we answered the question “how is cultured meat made?” in the most inclusive and collaborative way possible by gathered industry views on safety priorities from 50 companies.
In Phase II, we gathered insight from the scientists and decision-makers within regulatory bodies tasked with performing safety assessments of these novel food products.
In Phase III, we are convening diverse stakeholders to create the infrastructure for shared knowledge, methods, and data that address the priorities identified in Phase I and II.
Our long-term goal is to coordinate and launch collaborative efforts to develop and validate analytical methods for CM safety analysis and use these methods to generate datasets that can be made publicly available to support transparent food risk assessments and policy-making processes.

Workshops
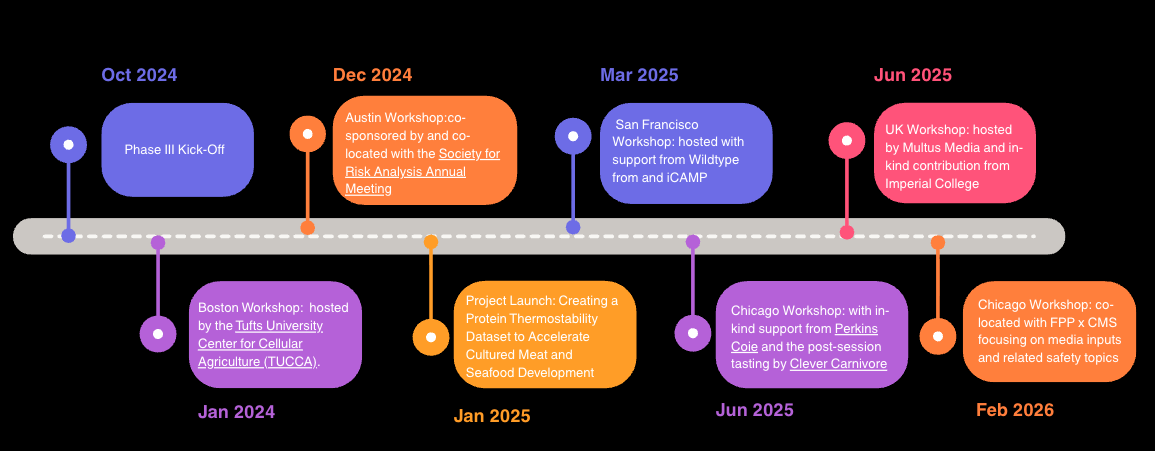
As a part of phase III, CMSI hosted four working sessions across the US; one session in London during Climate Week, and one in Amsterdam during the Future of Food Conference. These sessions are about bringing people together from academia, industry, government and NGOs, to coordinate efforts in developing open safety data and methods for cultured meat and seafood.
Over ninety-seven individuals from 94 organizations and 18 countries attended the 6 CMSI Phase III sessions to date, showing significant interest in collaborating to advance safety research.
Discussion topics:
-
Intentional genetic modifications & genetic drift
Novel allergens and toxins which could be produced by animal cells
Source animal health
-
Evaluating inputs (media ingredients, scaffolds, microcarriers, etc) for safety parameters
Stability of inputs and safety of potential metabolites
Residue levels in final products
Safety of media recycling
Assess whether media recycling concentrates hazardous inputs
Define how recycled media can be qualified to be safe
Threshold of Toxicological Concern (TTC) approach
-
Identification and risk assessment of common microbial contaminants
Adapt and validate testing methods
-
Study the shelf life of CM products and identify appropriate packaging/shipping conditions
Assess whether inputs or cooking methods impact bioavailability of nutrients in the final products
Develop and validate non-animal testing methods to demonstrate safety/conduct nutritional analyses (i.e. digestion, microbiome safety assessments, protein quality)
-
Evaluate safety of novel food processing techniques
Develop HACCP and prerequisite programs
Assessment of waste products and appropriate disposal procedures
Summary Reports
Funding and Support
Financial support for this working session series was provided by the US National Science Foundation (Grant No. 2417703) and the US Department of Agriculture National Institute of Food and Agriculture (AFRI project 2024-07959). Any opinions, findings, and conclusions or recommendations should not be construed to represent any official USDA or U.S. Government determination or policy.
In kind support was generously provided by The Society for Risk Analysis (SRA), Wildtype, the University of California Davis Integrative Center for Alternative Meat and Protein (iCAMP), Perkins Coie, Clever Carnivore, Multus Media, and The Bezos Centre for Sustainable Protein at Imperial College London.




