Phase II: International Regulatory & Government Perspectives
Gathering insights from governmental representatives around matters of safety for cultured meat.
Building off the industry insights gathered in CMSI Phase I, we convened 48 governmental scientists and regulators from 15 jurisdictions around the world. Through a series of interviews and workshops, we identified governmental priorities for the safety methods, data, and research needed to support safety evaluation of cultured meat products to reach commercial markets worldwide.

To advance cellular agriculture, it is critical to create public information to guide evidence-based policymaking and regulatory decisions.
In Phase I, we answered the question “how is cultured meat made?” in the most inclusive and collaborative way possible.
In Phase II, we aimed to gather insight from the scientists and decision-makers within regulatory bodies tasked with performing safety assessments of these novel food products.
Given how differently foods can be regulated across regions, it was an important opportunity to understand multiple perspectives to consider a best path forward for future research.
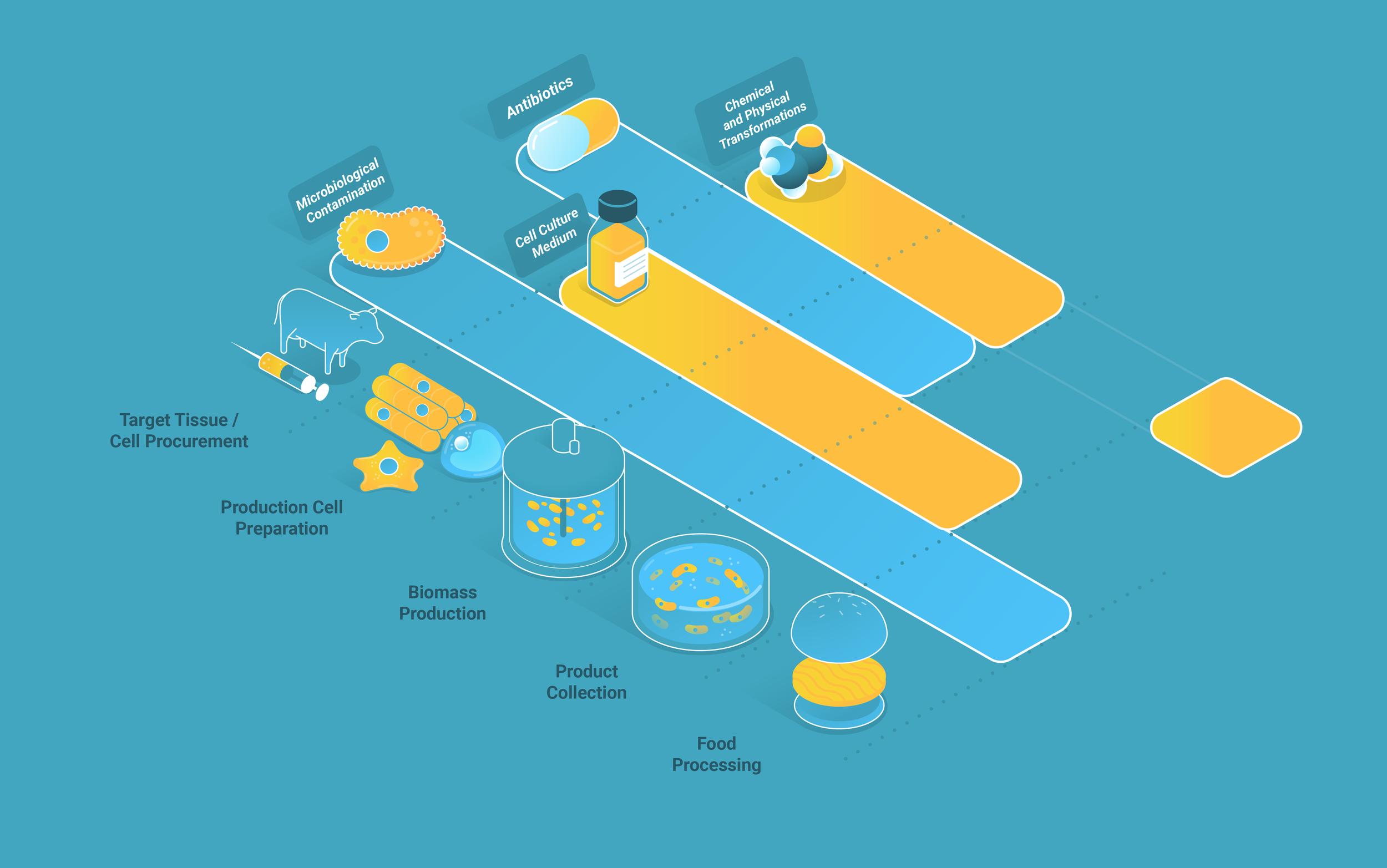
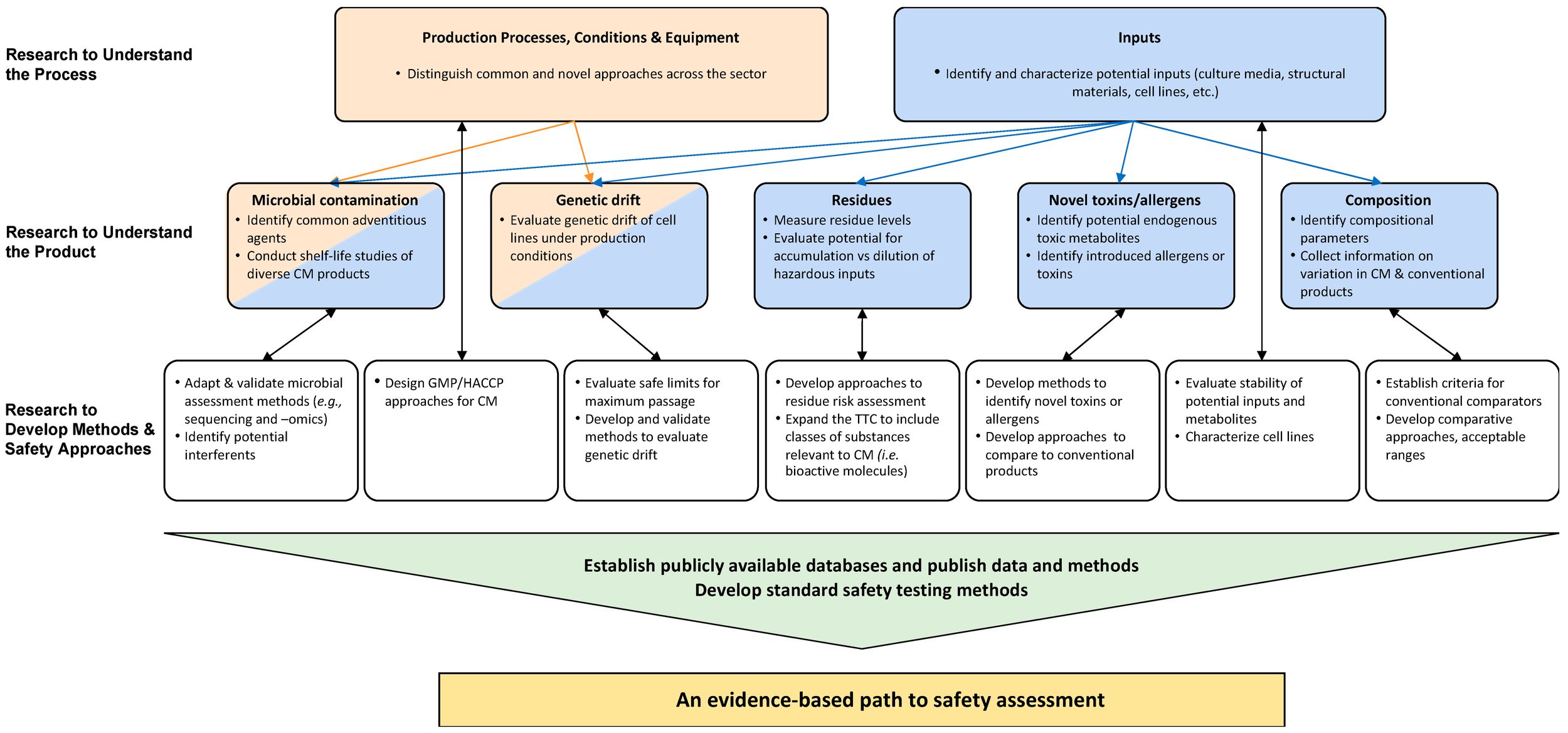
Figure 1. Safety assessment categories and checkpoints for the cellular meat cultivation process
Outcomes
Twenty interviews, two virtual workshops, and one in-person workshop culminated in a peer reviewed publication in Foods that identifies the key safety questions and priority areas of research going forward.
Figure 2. Research priorities identified by governmental scientists and researchers.
This key figure from the paper outlined an evidence-based path to safety assessment.
Funders and Support
This initiative was made possible thanks to the generous contributions of: Soroush Pour, Lejjy Gafour, Andras Forgacs, Gabor Forgacs, and Kristin Ellis, and to the 23 corporate donors listed below. Thank you to the Singapore Food Agency for hosting an in-person workshop in October 2022.